For every drug tablet or injectable solution on the market, countless hours were spent by the pharmaceutical companies to make sure that they are essentially free of particulate contamination.
When contaminants are encountered, FDA regulations require that the contamination problems be thoroughly investigated. For investigating particulate contamination issues, it is most important to document the size and color of the contaminants, identify the chemical composition of the contaminants, and locate their source of origin.
A pharmaceutical company received a batch of stoppers that had black particulate on them. The stoppers were to be used for an injectable solution.
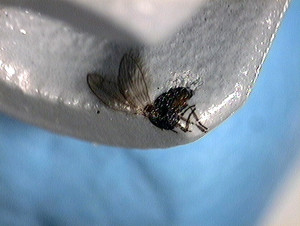
For particulate contamination identification, we begin the examination with a stereomicroscope to document the sample as we receive it. In this case, we could make out that the black particulate on one of the stoppers was a bug.
This was a different stopper examined at a higher magnification; you can clearly see the wings of the bug.
To remind you again, this was a black particulate contamination in a pharmaceutical stopper. The detail is so fine and surreal that it almost looks like the bug was printed on the stopper.
Since we have identified the black particulate to be a bug, the next question was whether the contamination got into the stopper at the pharmaceutical company or at the stopper manufacturing facility. To solve the mystery, we examined the stopper sample with a scanning electron microscope (SEM) to look at it at a higher magnification.
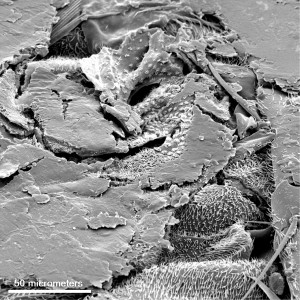
The first SEM image of the contamination shows that something is embedded in the stopper.
As we zoomed in, you can see that the bug parts are buried underneath the surface of the stopper.
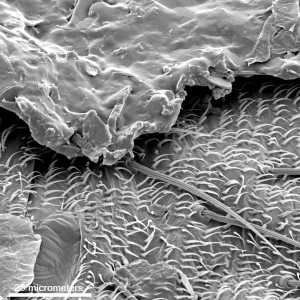
Another SEM view of the particulate contamination at a higher magnification clearly shows the hairs sticking out from the body of the bug underneath the surface of the rubber stopper.
Knowing that the bug was embedded in the rubber stopper, it was very unlikely that the contamination had occurred at the pharmaceutical facility. The contamination most likely happened during the stopper manufacturing process. With this information in hand, the pharmaceutical company was able to ask the stopper manufacturer to replace all the stoppers without any additional fees.
Click here to see our pharmaceutical testing capabilities.
Visit our Case Studies page for more stories!