Contamination is an ongoing battle for every pharmaceutical company. When pharmaceutical manufacturers encounter foreign material contamination in products, they have to quarantine the product and investigate and identify the origin of the contaminants. In this case study, a pharmaceutical manufacturer noticed streaks of foreign material in their vial caps. This was a big issue, and it would have caused major delays in fulfilling orders if they failed to identify the source of contamination.
MVA Scientific Consultants received the vials for foreign material identification. Upon close examination with a stereomicroscope, we noticed streaks of shiny metallic particles (Figure 1).

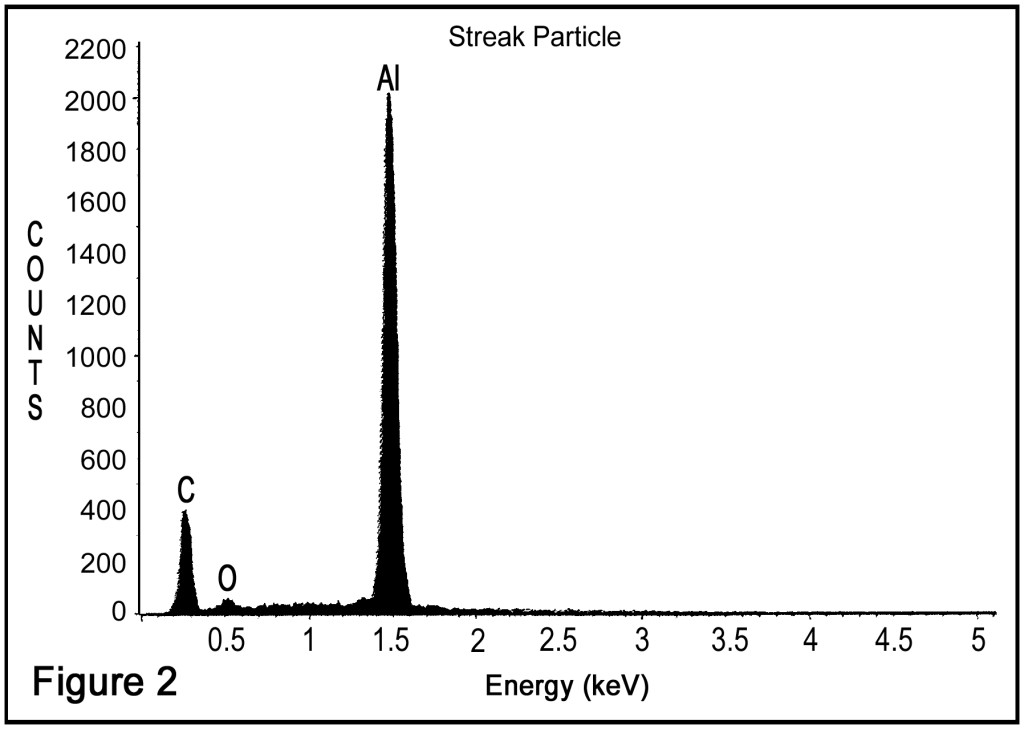
The foreign materials were comprised of micrometer-sized particles. The particles were not easily rubbed from the surface of the cap, and they did not appear to be abrasion or wear particles. Rather, they seemed to be a fine dust deposited on the caps. We cut out a section of the foreign material for scanning electron microscopy (SEM) analysis. The SEM showed that the particles are in the range of 20 to 50 micrometers. Elemental analysis by energy dispersive spectrometry indicated that the foreign materials were composed of elemental aluminum (Figure 2).
In conclusion, MVA’s analytical result suggested the aluminum particles were transferred to the caps from another surface. The pharmaceutical manufacturer was able to trace back to equipment in the production line that was responsible for depositing the aluminum streaks.




